Arrival of the Fittest: Solving Evolution's Greatest Puzzle (9 page)
Read Arrival of the Fittest: Solving Evolution's Greatest Puzzle Online
Authors: Andreas Wagner

Once metabolism and replication were in place, life was almost ready to crawl out of its cradle, but it still needed a travel bag. All of today’s life uses the same kind of material to pack up its molecules, lipid molecules that are
amphiphilic,
from the Greek words for “both” and “love.” An amphiphilic molecule “loves” both water and fat, because one of its ends likes to mingle with water, whereas the other avoids water—like oil that spreads in a thin film on a puddle. Observe lipid molecules in a solution and you are in for a surprise: They can form
vesicles,
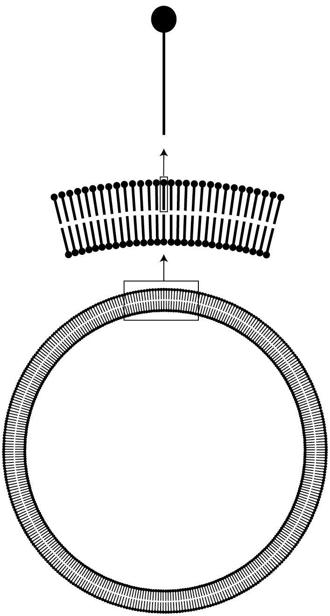
minute hollow droplets enclosed by a tiny spherical membrane, in which the lipid molecules are arranged, as shown in figure 2.
59
How they could arrange themselves into complex highly ordered membranes without a guiding hand may seem mysterious but is not that hard to understand: This arrangement satisfies both parts of each molecule. The water-loving parts (solid circles in the figure) are close to water, whereas the water-avoiding parts (sticks in the figure) are away from it and close to each other. What is more, these membranes can grow spontaneously, incorporating new lipid molecules as you add them to the solution. And they grow autocatalytically: The larger they are, the faster they can grow.
FIGURE 2.
Biological membranes
To see where the building blocks of these membranes came from, we do not have to look far. The citric acid cycle produces one of their precursors, and they arise even in extraterrestrial rocks like the Murchison meteorite. Heat up powdered meteorite with water, and you will find molecules that self-assemble into vesicles.
60
What is more, montmorillonite, the same vent mineral that can string together RNA, accelerates membrane assembly. And hydrothermal vents can help in further ways, by concentrating membrane ingredients. This is what a team around Jack Szostak from Harvard University found when they re-created tiny reaction chambers like those of hydrothermal vents in their laboratory. They heated small amounts of lipids in tiny capillaries and saw that the lipids become concentrated at one end, until they start to form vesicles.
61
All by themselves.
It smacks of Van Helmont’s spontaneous generation, all this complexity emerging from nothing but the right ingredients. But there is a crucial difference. Spontaneous generation—of mice, maggots, or microbes—requires the mysterious and perhaps supernatural vital force that Buchner’s discovery of enzymes began to expose as an old wives’ tale.
62
In contrast, the spontaneous creation of membranes and molecules—self-organization in modern language—requires only mundane physics and chemistry. Assembling membranes requires nothing but the attraction of similar molecules. Like the self-aggregation of volcanic particles into towering underwater buildings, or the spinning of RNA strings by clay minerals, the self-organization of both membranes and molecules is explained by well-known laws of nature.
Self-organization permeates the universe so completely that most of us don’t even notice it. Much older than life and natural selection, self-organization is how stars and solar systems form, how the earth accreted, how it acquired a moon, oceans, and an atmosphere, and how the continents started to shift. Self-organization creates the microscopic symmetry of a snowflake and the raging clouds of a hurricane, the shifting shapes of sand dunes and the timeless beauty of a crystal. We shouldn’t be surprised to find self-organization in life’s precursors, because it is everywhere else too.
Life’s self-organizing membranes can solve another one of early life’s puzzles: the mechanism by which the first cells divided. Modern cells use very sophisticated machinery—dozens of proteins—to constrict and divide cells, and to make sure that each daughter cell receives a copy of the mother’s DNA. But they could do the trick in simpler ways, as Szostak’s team found in 2009. The researchers observed how rapidly growing membrane droplets change their shape when they divide, and transform into threadlike hollow tubes. Unstable tubes, I should say. Agitate them a bit and they fragment spontaneously into smaller droplets. Even better, when the researchers placed RNA molecules inside these tubes, they were partitioned among the droplets. Lifeless membrane droplets can divide like living cells—an innovation without an innovator, emerging from a simple property of the system’s chemistry. All by itself.
Although we have come a long way from the first musings about a primordial soup, there are some problems that still defy solution. One of them is the last obstacle on the path from a self-dividing membrane droplet to a primitive cell. If the RNA inside this cell replicated faster than the cell grew, it would divide until the vesicle was ready to burst. But if the cell outpaced the RNA in growing, the RNA inside would become increasingly dilute, and many droplets would spawn empty-shelled offspring. To succeed, life needed to balance, to
regulate
replication and growth with precision, such that RNA replicated no faster than its container grew. How it learned to do that remains a mystery that twentieth-century science has left for another generation.
Fast-forward from the first wheel to the Ferrari. Although some of life’s features did not change in the thirty million centuries since it began—molecules, regulation, and metabolism are still wellsprings of innovation, as we shall see in later chapters—evolution transformed just about everything else about it. Early RNA replicators have been replaced by complex protein machines. Life has learned to regulate not just RNA and lipids but thousands of molecules. And innumerable innovations have turned the metabolism of a modern cell—the Ferrari’s engine—into a miracle of chemical technology.
Imagine driving home in this Ferrari from an evening picnic and running out of gas on a lonely stretch of highway in the middle of the night. No gas station is in sight, nor is anybody you could hitch a ride with. But no matter. You open your trunk, where a cooler contains leftover food and drinks. You pour a bottle of orange juice into the tank, and after it a quart of milk, and then a glass of wine. That will be enough to tide you over to the next gas station. And on you drive.
Modern metabolic engines are just like that. They can run on many different fuels. And more than that, they can also use each fuel as raw material to manufacture the smallest molecular parts of their body, parts the body needs to grow, to reproduce, and to heal. It’s as if a car could use the stuff in its gas tank not only to operate the engine but also to patch a leaky tire or mend a broken windshield.
The molecular parts in question comprise a few core molecules, some sixty
biomass building blocks
from which our bodies are constructed and repaired.
63
The most important are the four DNA building blocks of our genome, the nucleotides composed of a sugar, a phosphate group, and one of the four nitrogen-containing bases adenine (A), cytosine (C), guanine (G), or thymine (T). Next are the four building blocks of the RNA into which this DNA is transcribed, and that still controls much of life. They—A, C, G, and U, for uracil—differ only in a single oxygen atom from DNA building blocks, but this single atom makes a huge chemical difference. It makes RNA the better catalyst, and DNA the better—because more stable—information repository. Then there are the twenty amino acid building blocks of the amino acid strings translated from RNA, some of them familiar, like the tryptophan blamed for post–turkey dinner drowsiness, or the glutamic acid of the flavor enhancer monosodium glutamate (MSG). Together with the lipids in membranous bags, some energy storage molecules for hard times, and molecules that help enzymes do their job, these comprise the sixty different kinds of bricks from which cells build themselves.
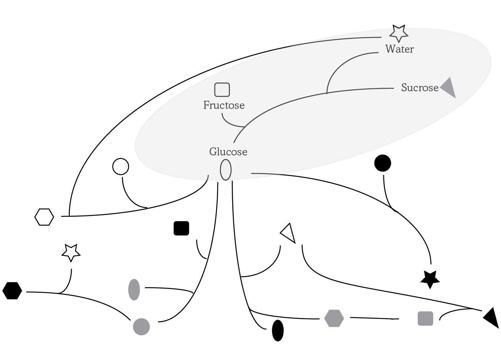
The tasks of metabolism—procuring energy and making stuff—have not changed in the last 3.8 billion years. And neither has its basic nature, a network of chemical reactions like the one where the white table sugar sucrose reacts with water and splits into the two more digestible molecules glucose and fructose. What has changed is the
number
of those reactions. Our earliest ancestors got by on a handful of reactions, but modern metabolism, like modern life in general, is much more complicated.
FIGURE 3.
A tiny sliver of a metabolic network
Modern metabolism is an interlaced, highly connected network of chemical reactions, the product of four billion years of innovation. If you were to chart it out, it would resemble a map of every street in the United States, from the shortest residential cul-de-sac to the complete interstate highway system. At its core is the ancient citric acid cycle—as central as Pennsylvania Avenue, which connects the White House with the U.S. Capitol. Figure 3 shows a tiny sliver of such a network, whose lines connect different molecules (shapes) that react with one another. Think of it as the road map of a village. The four molecules involved in cleaving white table sugar are written out and encircled within an ellipse. But don’t take this visual crutch for the real thing. Fructose can participate in thirty-seven reactions rather than the single one shown, and many more molecules and reactions are needed to run a modern metabolism.
To find out how many required more than a century of research. During this time, thousands of biologists built a tower of knowledge about metabolic reactions by studying the human gut bacterium
Escherichia coli.
Its construction took about as long as a medieval cathedral, but the vista from the top is spectacular. We now know how
E. coli
’s metabolism—more than a thousand small molecules that rearrange themselves in thirteen hundred metabolic reactions—is wired.
64
And we know that in the metabolism department
E. coli
and many other microbes beat us hands down. For example, of the twenty amino acids in our proteins, our bodies can only manufacture twelve. The other eight we have to get from food. In addition, we need thirteen vitamins to live, but can synthesize only two of them, vitamins D and B
7
(biotin).
65
E. coli
can cook all of them up from scratch.