Regenesis (5 page)
Authors: George M. Church

Sixth, at the extreme, a mirror cell or a mirror organism (composed of chemicals of reversed handedness) might be resistant to all or nearly all parasites and predators, a tremendously valuable result.
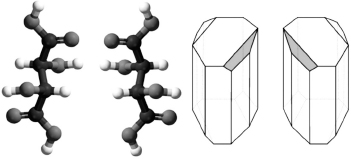
Since biomolecular handedness is so important, what is it? The basic idea is conveyed by the fact that our right and left hands are mirror images of each other and are not related by simple rotations. If we take a sculpture of a right hand and press it into a soft mold, we will discover that we cannot fit our left hand into the mold (
Figure 1.1
). However, if we fill that mold with plaster, the resulting new copies are considered complementary and are of the same handedness as the originals.
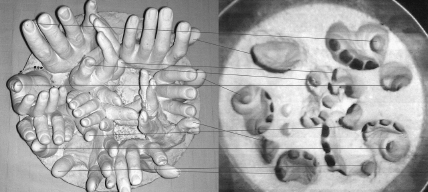
Figure 1.1
A sculpture on the left and corresponding negative moldâillustrating handedness and complementary shapes.
This same phenomenon exists on the molecular level. For example, there are two ways to arrange the four atoms that can bond to a carbon atom, and each will be a mirror image of the other. Furthermore, each will have predictably similar properties.
This left-right feature is also known as chirality, from the Greek (ÏείÏ) for “hand.” Even scientists who don't think about mirror worlds initially show great confusion as to whether the properties of mirror versions of molecules, cells, and bodies can be accurately predicted based on the properties of their nonmirror versions. Consider this. If you build a replica of an old-fashioned clock by only looking at its reflection, the copy will predictably tell time, but the numerals will be mirror images of the originals and the hands will rotate counterclockwise. These outcomes are precisely as anticipated.
Here's a simple demonstration that relates the hands and clock examples to molecules. Start with a central cantaloupe ball, and use toothpicks to successively place around it, in a clockwise order, a raisin, a piece of coconut, and a piece of nectarine all flat on the table. Then make another such structure using the same pieces of fruit but placing them counterclockwise. You can flip one over so that the two structures match, but if you add a bit of honeydew above and attached to the central cantaloupe, then no matter how you orient the structures you can still tell which was
clockwise originally and which wasn't. If you place them in front of a mirror you can see that they are each other's mirror image. Now let's replace fruit with atoms: H, CO, R, N. This is the general structure of an amino acid. The NH
2
is the amine and COOH is the acid. R refers to a “radical” (a group of atoms that behave as a unit) that varies with amino acid type.
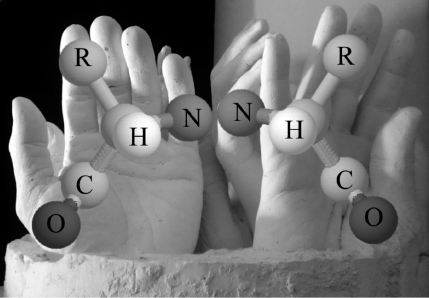
Figure 1.2
Another example of handedness uses a ball and stick model of a carbon atom with its four bonds (to H, CO, R, N groups).
Amino acids have a known handedness. You can impress your friends by your ability to identify the natural form. In nature, for reasons still unknown, almost all biomolecules vastly prefer one of the two hands (amino acids and proteins being designated as left-handed). Life itself, in a way, is fundamentally single-handed. Here is a procedure for telling whether a human hand, or a molecule, is right- or left-handed. Looking at your left hand palm up as in
Figure 1.2
, go from thumb to index finger to pinkie, the direction is clockwise, which indicates left-handedness. Performing the same observation on the right hand gives a counterclockwise direction, indicating right-handedness.
Now let's do the same for molecules. When looking down the bond from the hydrogen (H) to the central carbon, if the other groups going clockwise are CO, R, N, as on the left of
Figure 1.2
, then the configuration is normally seen in natural proteins (sometime called
levo
or L, for left-handed). On the right is the mirror version
(dexter
, Latin for “right,” or D). The R (radical) group distinguishes the twenty (or so) types of amino acids, each with its own personality (and its own single-letter code). Some are electrically negative while others are positive. Some are greasy and fear water (or hydrophobic), while others love water (hydrophilic). Glycine is the only amino acid that is its own mirror image, since its two hydrogen atoms are normally indistinguishable. Just to keep us on our toes, natural nucleic acids (RNA and DNA) were long ago designated D and their mirror forms L.
By now you may be wondering about the cash value of this talk about handedness. Just as there can be mirror molecules, there can also be mirror life. Mirror life would be the result of changing the handedness of an entire organism and all of its components, so that you have a mirror image of everything from the macro level all the way down to the atomic level. While mirror life may
look
identical to current life, it would be radically different in terms of its resistance to natural viruses and other pathogens. Mirror life forms would be immune to viruses and other pathogens, the reason being that the molecular interactions of life are exquisitely sensitive to the mirror arrangement of their component atoms and molecules. Normal viruses would not recognize a mirror organism as a genuine life form whose cells it could invade and infect. Such multivirus resistance would be an incredible boon to humanity. But it would come at a steep price because mirror life would be unable to digest foods by means of normal enzymes, which would mean that we would need to develop, cultivate, and mass-produce a whole range of mirror foodstuffs. (Although biohackers could in principle synthesize mirror viruses and other pathogens, mirror humans would still be resistant to natural pathogensâand even to genetically engineered nonmirror superpathogens.)
The prospect of mirror humans raises unusual and startling possibilities. Supposing that there will be a transition to a mirror version of human
beings at some point in the distant future, the changeover would be gradual, with a substantial interregnum period when two types of human beings would exist: natural humans composed of natural-handed molecules, and mirror humans made up of mirror versions of them. In this situation, it's almost as if two separate species of humans existed simultaneously. Or we might see an equilibrium between the two types if mirror pathogens arose.
These mirror humans should have an unusual smell. Members of the two versions could marry, but producing children would require what today would be considered extraordinary efforts. However, by the time we can make mirror humans, making designer (or random) children of either mirror type will not seem as challenging as it does today. We might even be able to make mirror identical twins or bodies that are mixtures of both types of cells.
Finally, creating a race of mirror humans is not without risks. Although new mirror molecules interact with mirror versions of existing molecules in predictable ways, how they interact with biomolecules in general is unpredictable. However, they are no more unpredictable than any newly synthesized drug, chemical, or material; nevertheless, careful screening of mirror molecules by computational methods or by actual experiment will be necessary to ensure safety.
Louis Pasteur had the first inklings into what natural chemical chirality is all about. He acquired this understanding by performing what the magazine
Chemical and Engineering News
once referred to as the “most beautiful chemistry experiment in history.”
Pasteur's seventy-two years on earth are remembered mainly for his contributions to microbiologyâespecially for inventing pasteurization, discovering the “Pasteur effect” (the anaerobic growth of organisms), developing the first vaccines for rabies and anthrax, and contributing to the understanding of fermentation, as well as for his clever experiments in support of the germ theory of disease. Nevertheless, his earliest and
equally great achievements came from his work as a crystallographer. In 1848, the seventy-three-year-old French physicist Jean Baptiste Biot sponsored the twenty-five-year-old Louis Pasteur in his first experiments at the elite college Ãcole Normale Supérieure in Paris.
This is a story about tartar, a chemical extracted from grapes. The modern chemical term “racemic” (meaning a mixture of the two hands) comes from the Latin
racemus
for “cluster of grapes.” In 1838 Biot found that tartaric acid, unlike its isomer, racemic acid, was optically active, meaning that it rotated the plane of a beam of polarized light. Both isomers are found in wine, the latter in sediments or by heating tartaric acid. These two acids were one of the first examples of an isomer pair and they turned out to be unusual in that almost all of their physical and chemical properties were identical except for their solubility and their ability to rotate polarized light.
Pasteur first showed that an equal mixture of the two forms of a salt of tartaric acid will spontaneously separate into small crystals. He heroically separated these microscopic crystals with tweezers and then dissolved them and showed that the solutions rotated polarized light in opposite directions. This key optical property is independent of orientations of the molecules, since in solution the molecules can take on all possible orientations. Furthermore, he made the profound observation that tartaric acid originating from natural systems (yeast from his wine making) came in only one of the two-handed states, the left-handed version. Since then we have learned that this propensity for single-handedness is a characteristic of most molecules that are constituents of living systems.
Omne vivum ex vivo
: “all life from life,” the irreducibility of living matter to anything nonliving, became one of Pasteur's most strongly held convictions. Indeed, Pasteur's key role in debunking the theory of “spontaneous generation of life” in 1859 could be construed as equally aimed at propping up the idea that the living forms of chemicals cannot be synthesized. That idea was consistently opposed by French chemist Marcelin Berthelot, who believed in the power of synthetic chemistry and experimentally synthesized organic substances that did not occur in nature. The dispute between Pasteur and Berthelot was not settled until 1971, when
H. P. Kagan synthesized the aromatic chemical helicene, a helical-shaped molecule, following Le Bel's 1874 proposed asymmetric synthesis using circularly polarized light.